FDA ends DCM updates; No causality data with dog foods

In 2022, the United States Meals and Drug Administration continued investigating cases of non-hereditary canine dilated cardiomyopathy, together with correlations with specific dog foodstuff and substances. Having said that, right after declining case studies, the company options to finish schedule updates on the investigation.
The Food and drug administration gained considerably fewer experiences of DCM conditions from 2020 to 2022 compared to the preceding two a long time. In overall, the Fda received 1,382 reports of DCM from Jan. 1, 2014 to Nov. 1, 2022. Even so, most of those case reports clustered about the dates of Food and drug administration announcements about its investigation of correlations among the grain-free doggy meals and DCM.
The Fda stated it had inadequate data to create causality among DCM circumstance studies and pet foods items eaten by troubled dogs.
“FDA does not intend to release further public updates right until there is significant new scientific details to share,” the company introduced in a push release on Dec. 23. “FDA has followed up on a subset of these studies, but is not able to examine every single report to confirm or confirm the documented info. Although adverse event numbers can be a prospective signal of an challenge with an Food and drug administration-controlled product or service, by themselves, they do not supply sufficient facts to establish a causal romantic relationship with described solution(s). Food and drug administration continues to stimulate investigate and collaboration by academia, veterinarians, and sector.”
Fda investigation into DCM and grain-totally free canine foodstuff
In July 2018, the Fda publicly announced the agency’s investigation into correlations amongst selected puppy foodstuff and DCM. Federal authorities examined experiences of DCM in pet dogs feeding on particular meal plans labeled as grain-free, specifically these containing peas, lentils, other legume seeds, or potatoes as principal substances, which had been far more frequent in meal plans labeled as grain-cost-free. A yr later on, the agency launched details from their investigation that stated 93{95221ed7c1b18b55d17ae0bef2e0eaa704ccc2431c5b12f9d786c88d1acb538d} of the 524 documented cases of DCM, included dog foods manufactured with peas and/or lentils, although 90{95221ed7c1b18b55d17ae0bef2e0eaa704ccc2431c5b12f9d786c88d1acb538d} of the troubled canines had eaten diets labeled as grain-cost-free. The Food and drug administration named 16 makes most usually eaten by puppies concerned in official reports of DCM. Product sales of grain-free pet dog food stuff in standard, and all those named makes in particular, fell following FDA’s announcements, even though more new items began to include taurine. Pet meals businesses adapted their marketing while building new puppy food items, treats and toppers exclusively meant to relieve pet owners’ fears of DCM. Instead of saying that all grains in pet foods ended up unfavorable, brands turned “grain friendly” as they started to include historic grains or other “healthy grains whilst continuing to malign traditional grains like wheat and corn.
Although the Fda investigation induced upheaval in the pet food sector, scientists and other concerned with the pet meals business have pointed out the deficiency of immediate proof connecting all those grain-free pet foods to DCM, due to the fact the to start with Fda announcement. Furthermore, some have criticized the Food and drug administration for going community with the investigation ahead of good evidence existed, specially looking at the adverse financial outcomes for canine food items models.
Canine dilated cardiomyopathy heart disorder
DCM impacts dogs’ heart muscle tissues. The disease outcomes in an enlarged coronary heart. As the heart and its chambers develop into dilated, pumping gets more tough and heart valves may perhaps leak, leading to a buildup of fluids in the upper body and stomach. DCM generally success in congestive coronary heart failure. Coronary heart function could enhance in circumstances that are not linked to genetics with suitable veterinary remedy and dietary modification if caught early. Breeds that are generally a lot more routinely afflicted by DCM include huge and large breed puppies, such as Terrific Danes, Boxers, Newfoundlands, Irish Wolfhounds, Saint Bernards and Doberman Pinschers. It is much less popular in smaller and medium breed dogs, apart from American and English Cocker Spaniels. Scenarios noted to the Food and drug administration involved Golden and Labrador Retrievers, Whippets, a Shih Tzu, a Bulldog and Miniature Schnauzers, as properly as combined breeds.
Overall canine DCM circumstance studies updated
The Food and drug administration also up to date its circumstance report information in its Questions & Solutions: FDA’s Get the job done on Prospective Brings about of Non-Hereditary DCM in Pet dogs
The subsequent is an excerpt from the up to date Food and drug administration Q&A linked to the DCM study. The order has been altered to target on thoughts most suitable to the pet food items business.
5. How lots of reviews of DCM has Fda received?
Due to the fact the agency’s very last general public communication on the investigation in 2020, Fda carries on to acquire reports of DCM diagnoses in canine, but at reduce levels than expert from 2018 through 2020 (see figure and table beneath). Some of the stories involve extra than 1 person animal from a single domestic. While Food and drug administration has adopted up on a subset of these studies, the agency is unable to examine every report to validate or validate the noted information.
More than time, Food and drug administration has published data about the number of reviews the agency has received. These communications have different in their articles – from time to time such as cats, as very well as canine, or only which include those people reports that Food and drug administration had verified with data from a veterinary cardiologist. In December 2022 as element of a regimen update to Congress, Food and drug administration compiled details on canine DCM experiences submitted to the company. Food and drug administration has opted to share that data right here for community recognition.
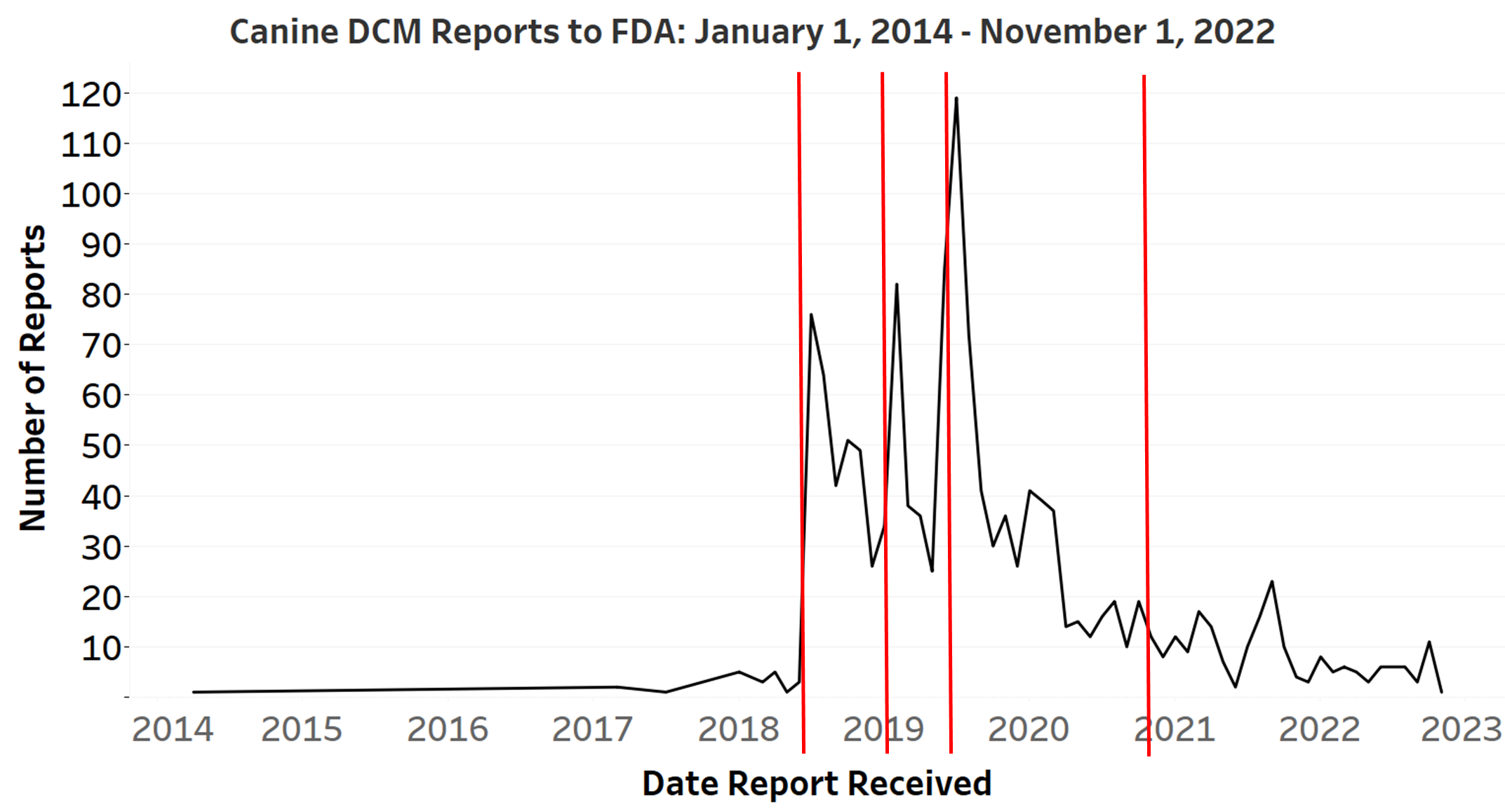
The graph shows the number of reports submitted to Food and drug administration considering the fact that 2014 and indicates the dates on which Fda issued general public updates on the investigation. The desk that follows demonstrates the variety of stories of DCM in dogs submitted in between past community communications from Fda, up to November 1, 2022.
A line graph of canine DCM reviews to Food and drug administration above the period of January 1, 2014 to November 1, 2022, slash by vertical strains exhibiting the dates that Fda launched general public communications about canine DCM on July 12, 2018, February 19, 2019, June 27, 2019, and November 3, 2020. The line graph displays sharp will increase in quantities of reports obtained shortly following the to start with 3 general public communications.
7. Is Fda organizing to continue naming the brand names most connected with instances?
No, we are not setting up to update the generally described makes, as we are mindful that a number of pet foods businesses have altered diet formulations considering the fact that our first bulletins about DCM. We have requested pet food manufacturers to share diet regime formulation facts, which could substantially profit our being familiar with of the position of diet in these scenarios. We continue to encourage pet homeowners to focus on their animals’ weight loss plans with their veterinarians.
1. What is canine dilated cardiomyopathy (DCM) and how does non-hereditary DCM vary from the genetic kind?
DCM is a ailment of a dog’s coronary heart muscle mass and effects in an enlarged coronary heart. As the coronary heart and its chambers turn out to be dilated, it turns into harder for the coronary heart to pump, and heart valves may possibly leak, which can guide to a buildup of fluids in the chest and abdomen (congestive heart failure).
Traditionally, DCM has been primarily connected to a genetic predisposition in certain breeds, but rising science seems to reveal that non-hereditary forms of DCM manifest in puppies as a advanced professional medical condition that may be impacted by the interplay of a number of factors this kind of as genetics, underlying medical ailments, and diet. Factors of diet that could interact with genetics and underlying health-related conditions might contain nutritional makeup of the components and how pet dogs digest them, component sourcing, processing, formulation, and/or feeding techniques.
Reports from veterinary cardiologists demonstrate some superior results in increasing heart operate in non-hereditary DCM scenarios, not like genetic varieties of DCM, with ideal veterinary cure and nutritional modification, when caught early in the development of the condition.
2. Why is Fda targeted on likely nutritional results in of non-hereditary DCM?
Whilst non-hereditary DCM seems to be brought on by a confluence of several elements, Food and drug administration is a regulatory company and has regulatory authority above animal foodstuff, such as pet food items, as a result the reason for the agency’s aim on food plan as a probable contributor. There is no public health and fitness company that tracks animal well being in the exact same way that the Facilities for Disorder Manage and Prevention tracks human well being, thus Fda has termed on the veterinary and tutorial communities, as well as sector, to contribute research on several facets of non-hereditary DCM. Lots of reps from these sectors participated in the new KSU scientific forum and several agreed to publicly share their abstracts and/or displays.
3. What is Fda executing to greater understand non-hereditary DCM scenarios?
Our veterinarians, animal nutritionists, epidemiologists and pathologists have been working with veterinary cardiologists and nutritionists from academia, sector and personal exercise to improved comprehend the clinical presentation of the cases and probable ties to diet regime, these as bioavailability of essential vitamins and how effectively a doggy digests these vitamins. Food and drug administration researchers also presentedExternal Hyperlink Disclaimer at the KSU symposium on a subset of puppies that experienced totally or partially recovered from DCM with food plan modify and veterinary treatment.
4. What more info would assistance further knowing of non-hereditary DCM?
Food and drug administration is encouraged by the response of veterinary cardiologists, veterinary nutritionists, academia and sector in delving into this concern and we really encourage other experts to acquire aspect. As we glimpse more into the position that eating plan might play in these conditions, we hope to take a look at added avenues of inquiry this sort of as formulation, nutrient bioavailability, component sourcing, and diet regime processing to figure out if there are any prevalent components. We have requested pet food stuff suppliers to share food plan formulation data, which could considerably advantage our comprehension of the job of food plan in the development of non-hereditary DCM. Formulation data shared with the Food and drug administration will be saved private.
Although quite a few of the reports submitted to Fda include things like considerable scientific information, such as echocardiogram benefits, cardiology/veterinary information, and comprehensive eating plan histories, the report counts are not limited to all those with substantial information and include things like all studies in which one or a lot more dogs was basically stated to have been diagnosed with DCM. Fda has adopted up on a subset of these experiences, but is not able to examine each individual report to validate or confirm the reported information. Food and drug administration did not include in the a lot of typical cardiac reviews submitted to Fda that did not report a DCM diagnosis.
It is usual for Food and drug administration to acquire a brief-time period raise in experiences after issuing community updates on a pet well being problem. In this case, upticks in the selection of DCM stories to Food and drug administration have a tendency to occur soon after Fda troubles general public updates on the DCM challenge. Community updates are indicated by the lines in the graph over.
In human health issues investigations, Fda is effective in live performance with the Facilities for Ailment Manage and Avoidance (CDC) and state boards of well being, which acquire and keep track of situations of foodborne illness. Sadly, there is no equal for pets, which usually means that it is complicated to accurately consider the scope of an animal ailment. For occasion, Food and drug administration is unaware of any figures on the qualifications amount of non-hereditary DCM in pet dogs. Reports to Fda are voluntary and probable do not reflect the legitimate incidence of the disease in the populace.
Fda continues to stimulate investigation and collaboration by academia, veterinarians, and market. Fda reviews all stories been given as section of our ongoing surveillance attempts relevant to pet food items basic safety. Even so, owing to source constraints the agency is not in a position to comply with up or present updates on each report.
6. Do the diet programs linked with instances of non-hereditary DCM show up to have any commonalities?
Most of the diet programs affiliated with the studies of non-hereditary DCM have legume seed elements, also identified as “pulses” (e.g., peas, lentils, and so on.), high in their component lists (while soy is a legume, we did not see a sign affiliated with this component). These involve equally “grain-free” and grain-containing formulations. Legumes, like pulse elements, have been applied in pet foods for many several years, with no proof to suggest they are inherently harmful, but evaluation of facts noted to CVM indicates that pulse ingredients are utilized in quite a few “grain-free” diets in greater proportion than in most grain-containing formulation. Food and drug administration has asked pet food items producers to give diet program formulations so we can further realize the proportions of ingredients in commercially-accessible weight loss plans and probable relationships with non-hereditary DCM.
The Food and drug administration does not know the distinct connection involving these diet programs and situations of non-hereditary DCM and is continuing to explore the part of genetics, underlying professional medical disorders, and/or other elements.
8. Is this an issue with only grain-absolutely free eating plans or meal plans made up of legumes or pulses?
No. Food and drug administration has been given stories of non-hereditary DCM linked with equally grain-cost-free and grain-containing weight loss plans. Most of the eating plans linked with reports of non-hereditary DCM have non-soy legumes and pulses (e.g., peas, lentils, etcetera.) superior in their ingredient lists. On the other hand, it is critical to notice that legumes and pulses have been utilised in pet foods for quite a few years, with no evidence to suggest they are inherently unsafe. CVM’s information present that pulse substances are probably employed in several “grain-free” diets in better proportion than in most grain-containing formulas.
9. As a regulatory agency, has Fda asked for any recollects of pet food items linked with non-hereditary DCM?
Fda has no definitive facts indicating that the eating plans are inherently unsafe and need to have to be removed from the market place, but we are continuing to do the job with stakeholders in examining how the diets could interact with other elements that may be impacting non-hereditary DCM. We really encourage pet proprietors to work with their veterinarians, who may well seek the advice of with a board-qualified veterinary nutritionist, prior to earning diet program alterations. We also encourage veterinarians to evaluate the proceedings of the KSU symposium in get to discover much more about non-hereditary DCM. Food and drug administration continues to pursue scientific being familiar with of non-hereditary DCM and welcomes added contributions from other experts.
10. How lengthy will it choose to pinpoint the lead to(s) of non-hereditary DCM?
We see this as an ongoing, collaborative scientific enterprise, of which Food and drug administration has just 1 piece as the regulator of animal food items and reviewer of adverse event stories received as portion of the pet foodstuff early warning and surveillance procedure. The scientific neighborhood continues to assess the out there data and fill data gaps to determine what elements may lead to the development of non-hereditary DCM. We search ahead to continuing to interact with scientists as opportunities arise. We will also supply further updates to notify the general public if or when substantive scientific data co






